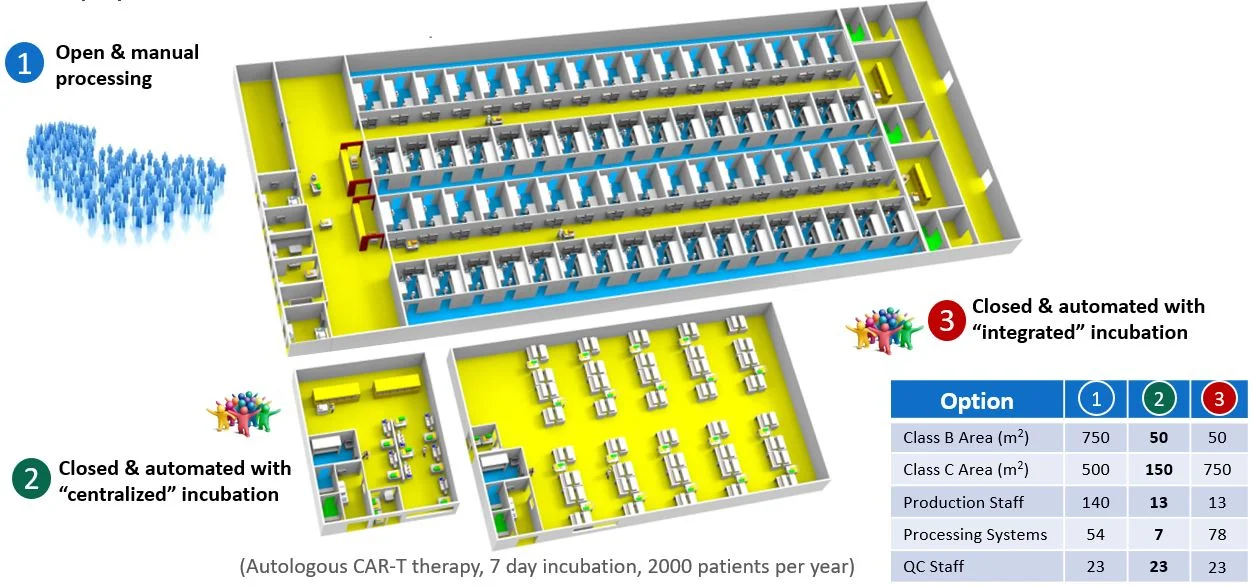
Cell therapy companies often inadvertently compromise commercial success because they fail to adequately explore or embrace alternative manufacturing technologies. The reasons are varied including:
- The possibility that existing clinical data will be put at risk by making a process change
- Clinical trials are already defined or approved based on the existing process
- The cost, quality and operational difficulties associated with scaling up the current process are not understood
- They are unaware of alternative technologies or technologies are simply not available
- There are more urgent priorities for staff and precious capital
However, with an experienced cross-functional team and careful planning, an appropriate scale-up strategy can be created, regardless of where you are in your clinical trials.
Ask us how.